GSECARS
A national user facility for frontier research in the earth sciences using synchrotron radiation at the Advanced Photon Source, Argonne National Laboratory.
GSECARS provides earth scientists with access to the high-brilliance hard x-rays from this third-generation synchrotron light source. All principal synchrotron-based analytical techniques in demand by earth scientists are being brought to bear on earth science problems.
SEES Multigrain Single Crystal Workshop
University of Chicago
August 8-9, 2026
Upcoming Events
Recent Publications
Mitigating lead bioaccessibility in metal (loid) s contaminated alkaline soils using soil amendments: Evaluating efficacy using in vitro bioaccessibility, sequential extraction, and synchrotron techniques
Wekumbura, Chandima, Ganga M. Hettiarachchi, William Hargrove, Qing Ma, Matthew Newville, Antonio Lanzirotti, and Christina Sobin. "Mitigating lead...
Synchrotron X-rays elucidate the intracellular storage of the iodide and bromide antioxidant system in the kelp Laminaria digitata
Frithjof C. Küpper, Mijovilovich Ana, Peter Cloetens, Peter M. H. Kroneck, Matthew Newville, Antonio Lanzirotti, Eric P. Miller, Martin C. Feiters,...
Complex redox histories of lunar pyroclastic beads revealed by spatially correlated chromium and Vanadium valences
Sutton, S. R., Lanzirotti, A., Newville, M., Dyar, M. D., McCanta, M., & the ANGSA Team (2026). Complex redox histories of lunar pyroclastic...
Controls on iron‐redox state in martian magmas quantified by Mössbauer spectroscopy, colorimetric wet chemistry, and XANES spectroscopy
Aithala, S. P., R. A. Lange, and M. M. Hirschmann. "Controls on iron‐redox state in martian magmas quantified by Mössbauer spectroscopy,...
Bulk superconductivity up to 96 K in pressurized nickelate single crystals
Feiyu Li, Zhenfang Xing, Di Peng, Jie Dou, Ning Guo, Liang Ma, Yulin Zhang, Lingzhen Wang, Jun Luo, Jie Yang, Jian Zhang, Tieyan Chang, Yu-Sheng...
Thermal evolution of the sulfur‐rich, small terrestrial planetary core as inferred from the experimental study of the Fe‐S‐O‐H system
Lee, Jeongmin, Jaeyoon Keum, Taehyun Kim, Chanbi Lee, Stella Chariton, Vitali Prakapenka, Nico Giordano, Byungdal So, Huijeong Hwang, and Yongjae...
Science Highlights
Mitigating lead bioaccessibility in metal (loid) s contaminated alkaline soils using soil amendments: Evaluating efficacy using in vitro bioaccessibility, sequential extraction, and synchrotron techniques
Wekumbura, Chandima, Ganga M. Hettiarachchi, William Hargrove, Qing Ma, Matthew Newville, Antonio Lanzirotti, and Christina Sobin. "Mitigating lead...
Synchrotron X-rays elucidate the intracellular storage of the iodide and bromide antioxidant system in the kelp Laminaria digitata
Frithjof C. Küpper, Mijovilovich Ana, Peter Cloetens, Peter M. H. Kroneck, Matthew Newville, Antonio Lanzirotti, Eric P. Miller, Martin C. Feiters,...
Complex redox histories of lunar pyroclastic beads revealed by spatially correlated chromium and Vanadium valences
Sutton, S. R., Lanzirotti, A., Newville, M., Dyar, M. D., McCanta, M., & the ANGSA Team (2026). Complex redox histories of lunar pyroclastic...
Controls on iron‐redox state in martian magmas quantified by Mössbauer spectroscopy, colorimetric wet chemistry, and XANES spectroscopy
Aithala, S. P., R. A. Lange, and M. M. Hirschmann. "Controls on iron‐redox state in martian magmas quantified by Mössbauer spectroscopy,...
Bulk superconductivity up to 96 K in pressurized nickelate single crystals
Feiyu Li, Zhenfang Xing, Di Peng, Jie Dou, Ning Guo, Liang Ma, Yulin Zhang, Lingzhen Wang, Jun Luo, Jie Yang, Jian Zhang, Tieyan Chang, Yu-Sheng...
Thermal evolution of the sulfur‐rich, small terrestrial planetary core as inferred from the experimental study of the Fe‐S‐O‐H system
Lee, Jeongmin, Jaeyoon Keum, Taehyun Kim, Chanbi Lee, Stella Chariton, Vitali Prakapenka, Nico Giordano, Byungdal So, Huijeong Hwang, and Yongjae...
Viscosity and structure of hydrous silicate liquids: Constraints on oceanic hydrothermal circulation
Chen, Qi, Craig Lundstrom, Yanchong Li, Young Jay Ryu, Tony Yu, Stella Chariton, Dongzhou Zhang, Vitali Prakapenka, and Yanbin Wang. "Viscosity and...
The Impact of Sample Size on X-Ray Microtomography Measurements of Fluid–Fluid Interfacial Areas
Araujo, Juliana B., Kieran McDonald, Kenneth C. Carroll, and Mark L. Brusseau. 2026. "The Impact of Sample Size on X-Ray Microtomography...
Pyrite (001) interface chemistry is controlled by a sulfoxy termination
Wanhala, Anna K., Piotr Zarzycki, Sergio Carrero, Michael Whittaker, Slavomir Nemsak, Yarong Qi, Joanne E. Stubbs, Peter J. Eng, and Benjamin...
Widespread Occurrence of Magnéli Phases in Wildland–Urban Interface Fire Ashes
Baalousha, Mohammed, Morgane Desmau, Roberto A. Colina-Ruiz, Antonio Lanzirotti, Sheryl A. Singerling, Michelle A. Stern, and Charles N. Alpers....
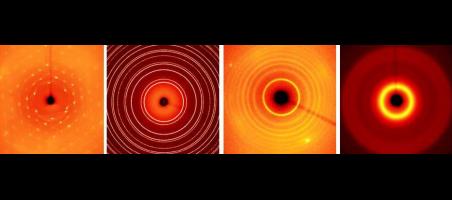
Equation of state and structural variations of synthetic forsterite-fayalite solid solution by single-crystal X-ray diffraction
Ye, Z., Xu, J., Zhang, D. et al. Equation of state and structural variations of synthetic forsterite-fayalite solid solution by single-crystal X-ray...
Latest Happenings at GSECARS
13 ID-D Table Upgrade

Dongzhou Zhang at UC Lab School
GSE scientist Dongzhou Zhang spoke to young students at UC Lab School about...

Shradhanjli Ravikumar at 13 BMD
Shradhanjli Ravikumar (Harvard University) studies technical development for...

Yanbin Wang at the APS 2026 User Meeting
Yanbin Wang (GSECARS) presents "High Pressure Deformation in LVP Presses" at...

Sean Shieh at 13 BMD
Sean Shieh (University of Western Ontario) conducts a high-pressure brillouin...

Mark Rivers, Yanbin Wang, Dongzhou Zhang and Tony Yu (GSECARS) participated in the ISRD Workshop, Integration of Tomography and Diffraction Techniques for Rock Mechanics: Critical Challenges and Needs
GSE staff host a tour of the LVP Program at GSECARS for the ISRD Workshop....